Structural Characterization and Physicochemical Stability Profile of a Double Mutant Heat Labile Toxin Protein Based Adjuvant
Abstract
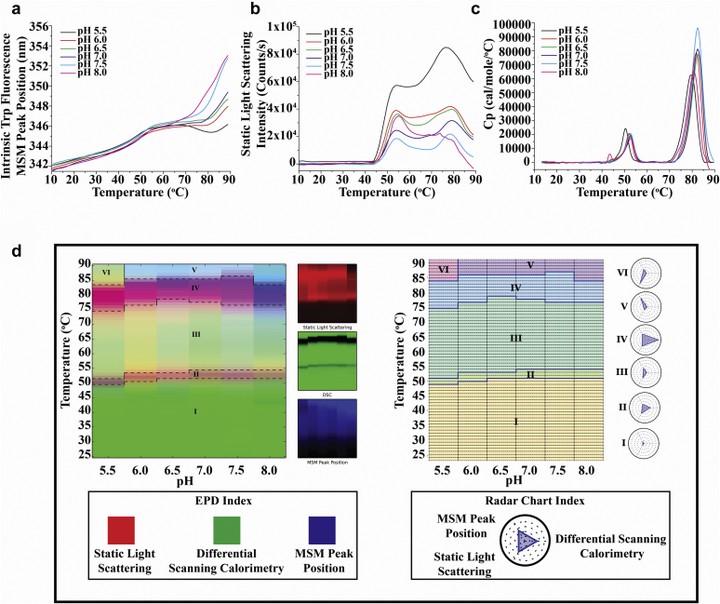
A novel protein adjuvant double-mutant Escherichia coli heat-labile toxin, LT (R192G/L211A) or dmLT, is in preclinical and early clinical development with various vaccine candidates. Structural characterization and formulation development of dmLT will play a key role in its successful process development, scale-up/transfer, and commercial manufacturing. This work describes extensive analytical characterization of structural integrity and physicochemical stability profile of dmLT from a lyophilized clinical formulation. Reconstituted dmLT contained a heterogeneous mixture of intact holotoxin (AB5, ∼75%) and free B5 subunit (∼25%) as assessed by analytical ultracentrifugation and hydrophobic interaction chromatography. Intact mass spectrometry (MS) analysis revealed presence of Lys84 glycation near the native sugar-binding site in dmLT, and forced degradation studies using liquid chromatography-MS peptide mapping demonstrated specific Asn deamidation and Met oxidation sites. Using multiple biophysical measurements, dmLT was found most stable between pH 6.5 and 7.5 and at temperatures ≤50°C. In addition, soluble aggregates and particle formation were observed upon shaking stress. By identifying the physicochemical degradation pathways of dmLT using newly developed stability-indicating analytical methods from this study, we aim at developing more stable candidate formulations of dmLT that will minimize the formation of degradants and improve storage stability, as both a frozen bulk substance and eventually as a liquid final dosage form.